How to Critically Appraise a Study: CASP and Risk of Bias in 15 Minutes
Reading a paper is easy. Trusting it is harder. Critical appraisal is the skill that helps you decide whether results are valid, biased, exaggerated, or applicable to your patients. This guide shows a practical way to appraise almost any clinical study in about 15 minutes.
If you are building your own research workflow, these posts connect well: How to Get a Research Idea, How to Write a Research Protocol, and How to Build a PubMed Search Strategy.
The 15-minute appraisal framework
- What question is the study answering? Identify population, exposure or intervention, comparator, outcomes, and timeframe.
- Is the design appropriate? RCT for causality, cohort for prognosis and associations, case control for rare outcomes, cross sectional for snapshots.
- Is there risk of bias? Randomization, allocation concealment, blinding, missing data, selective reporting, confounding.
- How big is the effect? Effect size and precision matter more than p-values.
- Can I apply this to my setting? Population, resources, baseline risk, and context.
Step 1: Read the study question like a reviewer
Write the question in one sentence using PICO:
- P: population
- I: intervention or exposure
- C: comparison
- O: outcome
If the paper does not clearly define P, I, C, and O, that is your first warning sign.
Step 2: Check validity and methods before results
Most readers jump to results. Reviewers do the opposite. Use these quick checks:
- Eligibility criteria: are inclusion and exclusion rules clear and reproducible?
- Outcome definitions: are endpoints defined precisely, with a clear time window?
- Measurement quality: were outcomes measured the same way in all groups?
- Follow-up: was it long enough and similar across groups?
Step 3: Risk of bias, explained visually
Bias is any systematic error that pushes results in one direction. Use the right tool for the design:
- Randomized trials: RoB 2
- Non-randomized interventions: ROBINS-I
- Diagnostic accuracy: QUADAS-2
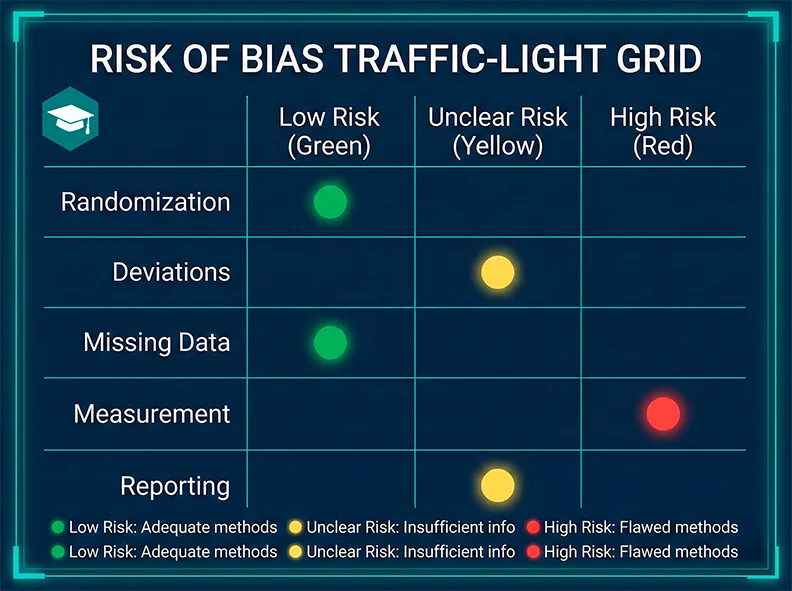
Authoritative references: Cochrane Library, EQUATOR Network.
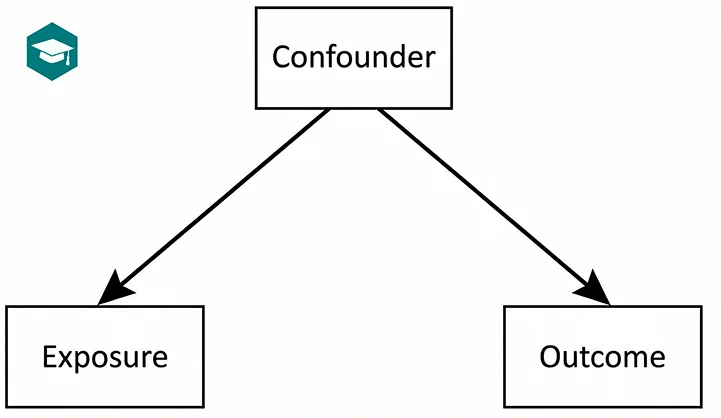
Confounding: the most common trap in observational studies
Confounding happens when a third factor affects both the exposure and the outcome. A paper can look convincing but still be wrong if confounding is not handled properly (matching, regression, stratification, inverse probability weighting, sensitivity analyses).
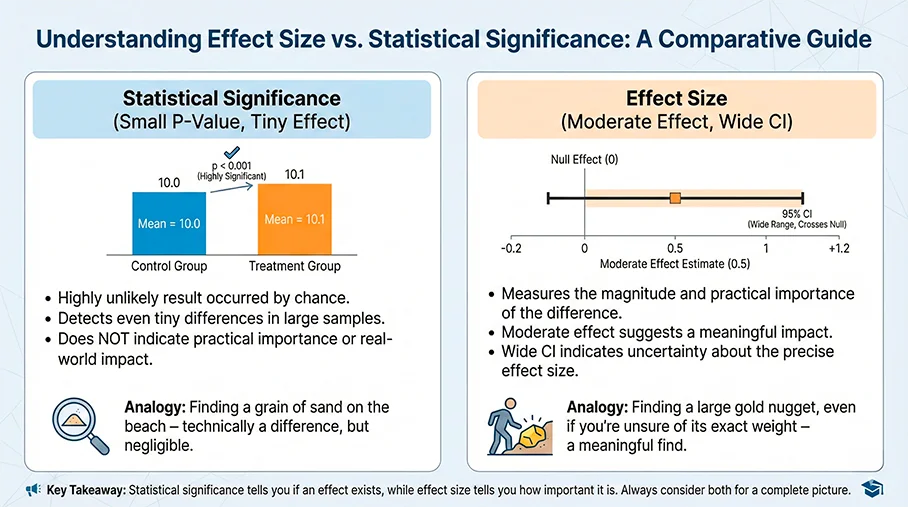
Step 4: Effect size beats p-values
Ask these questions:
- What is the effect size? risk ratio, odds ratio, hazard ratio, mean difference, or absolute risk difference.
- How precise is it? confidence interval width matters.
- Is it clinically meaningful? small statistically significant effects can be clinically trivial.
Step 5: Applicability to your setting
Even a low-bias study may not apply to your population. Check:
- Population match: age, comorbidity, severity, baseline risk
- Intervention realism: dose, operator skill, resources available
- Outcome relevance: patient-centered outcomes and meaningful follow-up
CASP-style checklist you can reuse (quick)
- Is the research question focused and answerable?
- Is the design appropriate for the question?
- Are the methods valid and clearly described?
- Is risk of bias low or addressed properly?
- Are results presented with effect size and confidence intervals?
- Are results applicable and clinically meaningful?
Outbound reference: CASP Tools and Checklists.
How to document appraisal in your workflow
If you are running a systematic review, store your appraisal decisions per study and per domain, then summarize them in the manuscript. In SciTrack, keep it inside your review workspace: Systematic Reviews Workspace.
For reporting, follow design-specific guidelines (CONSORT for trials, STROBE for observational studies). Outbound references: CONSORT and STROBE.
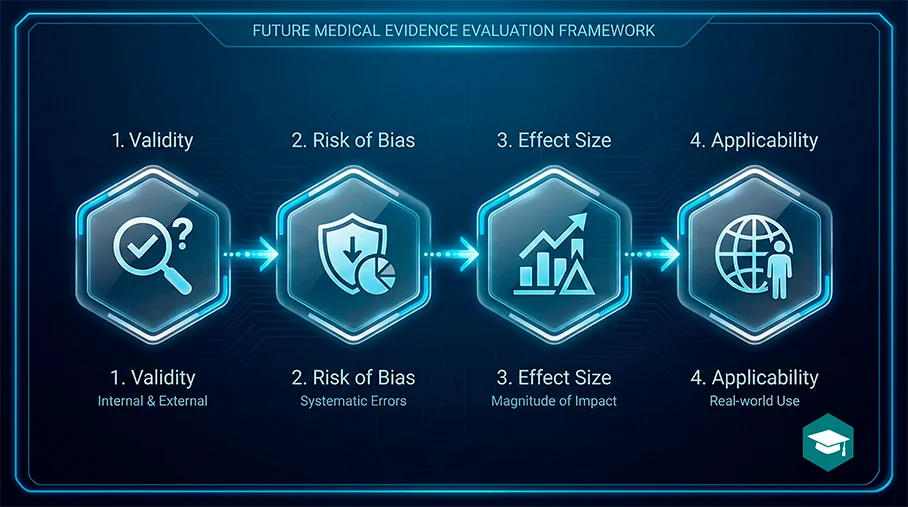
Conclusion
Critical appraisal is a repeatable skill. Start with the question, confirm validity, assess risk of bias, interpret effect sizes, and judge applicability. When you appraise studies consistently, your research decisions become faster, safer, and more publishable.




Comments (0)
Want to share your thoughts? Please sign in to leave a comment.
Sign In to CommentNo comments yet
Be the first to share your thoughts and start the discussion!